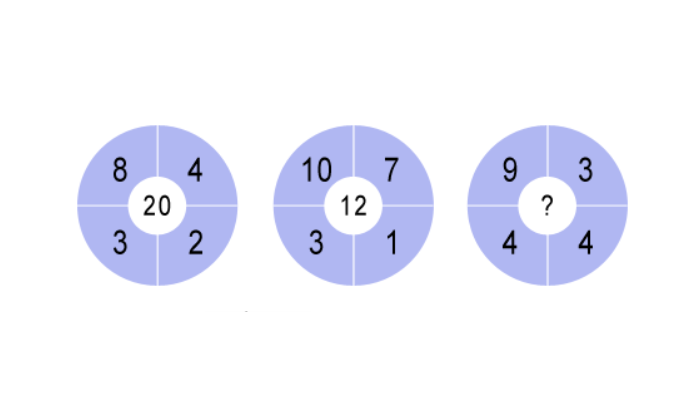
In the following circles, th...
In the following circles, the four outer numbers are used to calculate the number in the middle. Can you figure out the rule to determine what number should replace the question mark in the third circle?Correct answers: 108
The first user who solved this task is Jakubovski Vladimir.
#brainteasers #math #riddles