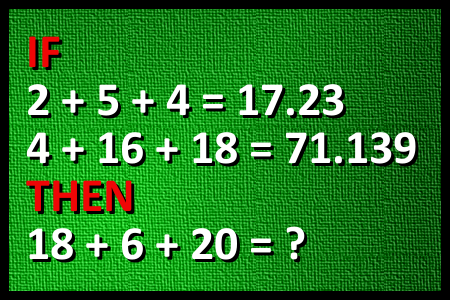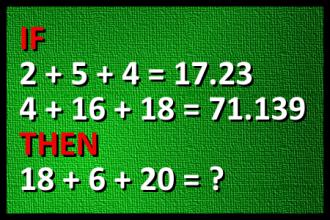Calculate 18+6+20
FUNNY MATH: Calculate 18+6+20 (Author: Ruzica Pavlovic)Correct answers: 25
The first user who solved this task is Sanja Šabović.
#brainteasers #math

Name the animals...
The first-grade teacher was showing pictures of animals to her students to see how many they could name. She held up a picture of a lamb, and a little girl said, "That's a sheep!"
"That's right!" said the teacher. "How about THIS one?" she said, holding up a picture of the king of beasts.
"That's a lion!" answered a little boy.
"Right!" said the teacher. Then she held up a picture of a deer. No one volunteered an answer. She tried to help. "What does your mother call your father?"
Johnny said, "I know! That's a lazy old goat!"