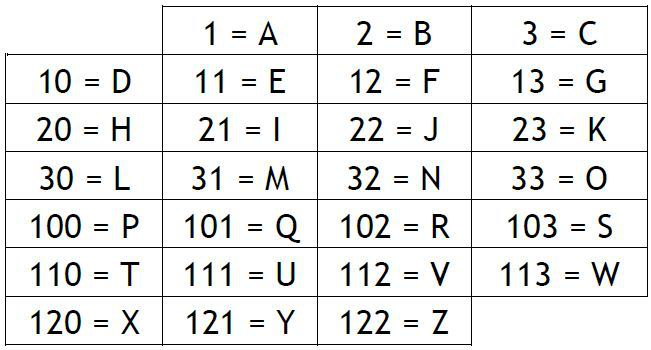
I have taken a quotation, an...
I have taken a quotation, and I have replaced each of the letters with one-, two- or three-digit numbers according to the table below. Can you change it back to letters? (2112 1110 1221102103110 12133111 103332110 10311133111110 313030 10110)Correct answers: 39
The first user who solved this task is llewellyn samuels.
#brainteasers #wordpuzzles #riddles

A woman walked up to a little...
A woman walked up to a little old man rocking in a chair on his porch.
"I couldn't help noticing how happy you look," she said. "What's your secret for a long happy life?"
"I smoke three packs of cigarettes a day," he said. "I also drink a case of whiskey a week, eat fatty foods, and never exercise."
"That's amazing," the woman said. "How old are you?"
"Twenty-six," he said.
"I couldn't help noticing how happy you look," she said. "What's your secret for a long happy life?"
"I smoke three packs of cigarettes a day," he said. "I also drink a case of whiskey a week, eat fatty foods, and never exercise."
"That's amazing," the woman said. "How old are you?"
"Twenty-six," he said.