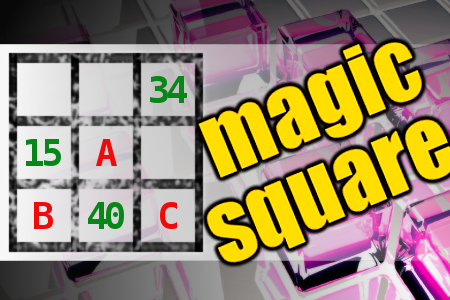
MAGIC SQUARE: Calculate A*B-C
The aim is to place the some numbers from the list (8, 12, 15, 16, 27, 31, 33, 34, 37, 40) into the empty squares and squares marked with A, B an C. Sum of each row and column should be equal. All the numbers of the magic square must be different. Find values for A, B, and C. Solution is A*B-C.Correct answers: 15
The first user who solved this task is Nílton Corrêa de Sousa.
#brainteasers #math #magicsquare